| |
| Unusual internal substrate binding and activation |
| |
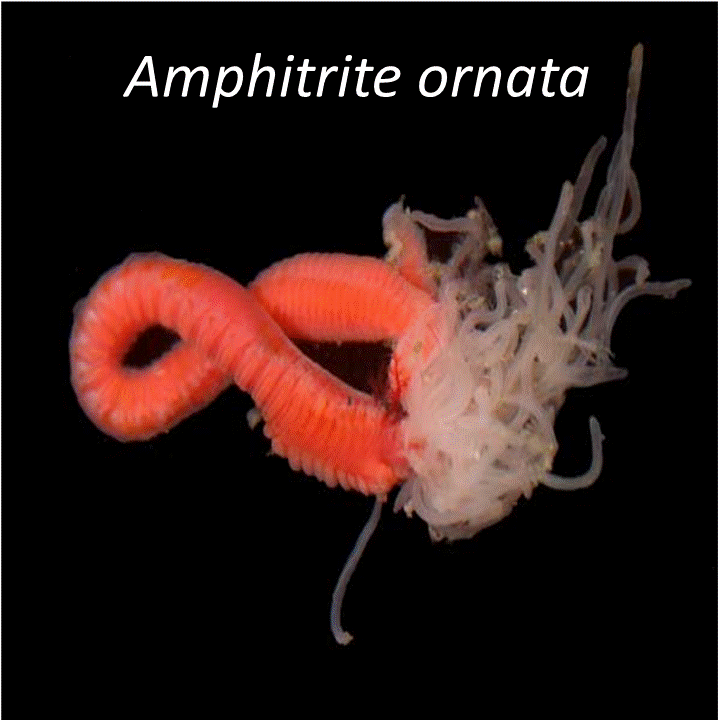
Although Amphitrite ornata was originally discovered as a hemoglobin in 1977 by Bonaventura's group at Duke in 1977, the protein was rediscovered as a dehaloperoxidase in 1996 by Lincoln's group at the University of South Carolina. The structure of a hemoglobin that acts as a peroxidase would have perhaps not received such great attention if it were not for the unusual mode of substrate binding inside the distal pocket of the protein. Hemoglobins will only bind large molecules inside the protein if those molecules form chemical bonds to the heme iron. DHP binds many molecules and our group has studied modes of binding and inhibition. Subsequently, our collaborator Dr. Ghiladi (also at NC State University) discovered new functions beyond the peroxidase function. The discoveries began with the realization that 4-bromophenol can bind internally and act as an inhibitor of peroxidase activation of substrates that bind externally.
Fluoride binding as a thermodynamic assay for substrate binding
Kinetics of peroxidase substrates and hydrogen peroxide
| |
|
|